Research activities Kluger-Lab
The main focus of research is on tissue engineering, such as the reproduction of human skin in order to create an alternative to animal testing. In addition, the Kluger Lab is investigating how real meat can be grown from cells in the laboratory without any animal suffering. The focus here is on production processes such as 3D printing.
Current research projects
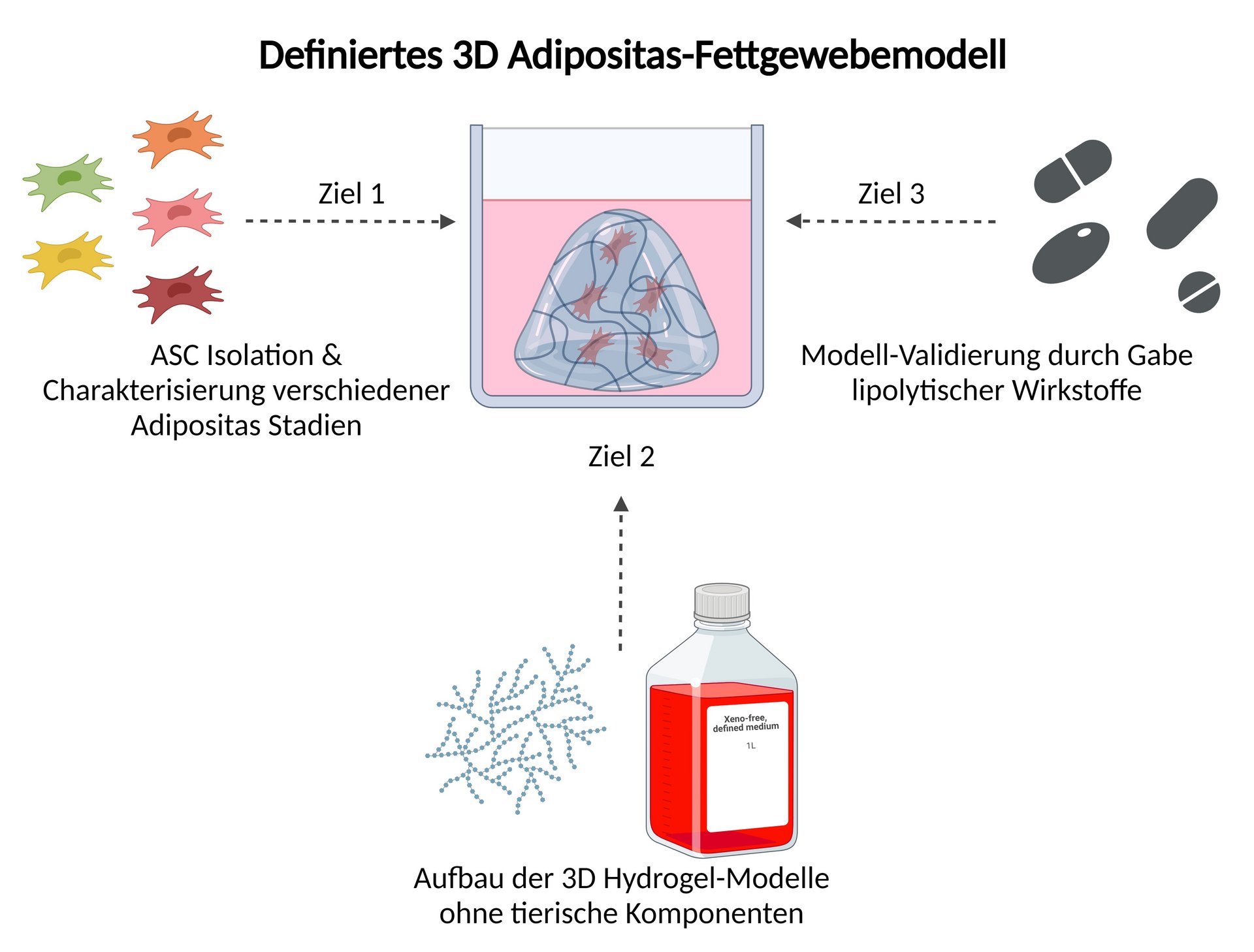
The Adiposity 3D project aims to develop a human 3D adipose tissue model without animal components. This hydrogel-based model will be constructed using primary stem cells from obese donors and will be used to test lipolytic agents to confirm the functionality of the in vitro model as an alternative to animal testing.
The objectives of the project are
- The isolation of primary human adipose-derived stem cells (ASCs) from obese and non-obese donors and the classification and characterization of the isolated cells based on their disease stage.
- Construction of in vitro human 3D models, without animal components, using the characterized cells and verification of cell status
- Validate the 3D models by adding lipolytic agents and studying the effect on the corresponding obesity states.
Project duration: 12/2023, for 2 years
Contact: Petra Kluger, Franziska Albrecht
INVERS – In vitro production of smart fish proteins: Establishment of fat precursor cells from salmon-like fish for the development of sustainable, high-quality fish foods
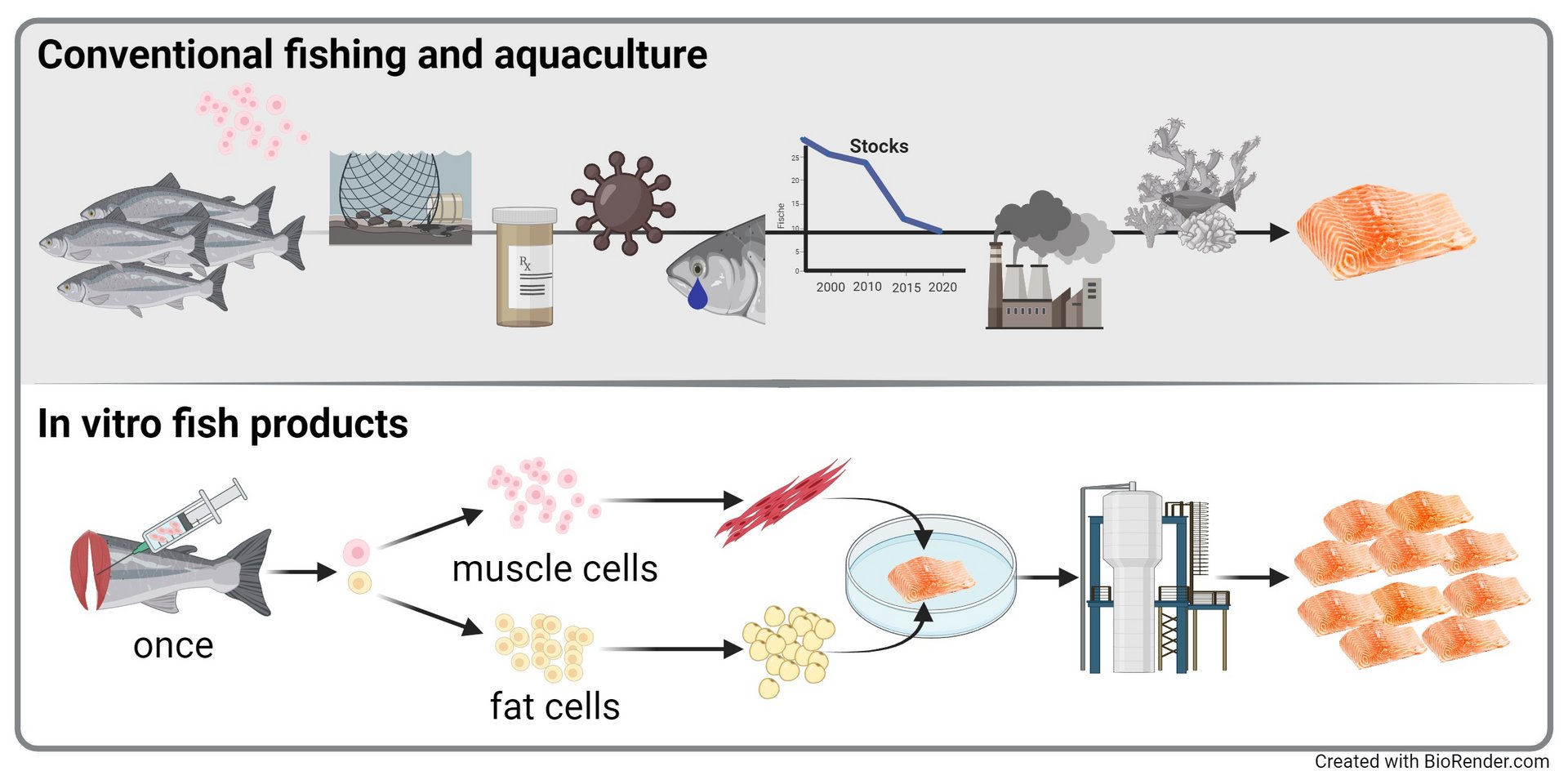
Fish products are an integral part of the human diet. Approximately 156 million tonnes of fish were consumed globally in 2018, which represents a doubling of consumption over the last 60 years to an estimated 20.5 kg per person. Fish products are an important source of nutrients (FAO 2020). This is particularly true for 'oily' fish such as salmon, trout and mackerel, which contain high levels of polyunsaturated omega-3 fatty acids in their fat tissue. However, a permanent supply of healthy fish products is not guaranteed. More than 30 % of the world's fish stocks are considered over-exploited, and a further 60 % are fished to their limits (FAO 2020). In addition, fatty fish in particular tend to store persistent environmental toxins such as mercury and dioxins in their tissues. New approaches are therefore needed to ensure a sustainable supply of healthy seafood, especially as the farming of fish in aquacultures - as an alternative to wild-caught fish - is associated with a number of negative environmental impacts and the farming conditions are questionable from an animal welfare perspective.
In vitro technology is a new approach to develop alternative protein sources that can contribute to a sustainable supply of high-quality fish products. The first step in this approach is to remove tissue from a fish. The next step is to extract progenitor or stem cells from the fish tissue, which are then multiplied under controlled conditions. The cells are fed with nutrients in bioreactors. The cell biomass can then be harvested for the production of unstructured fish tissue. The following diagram compares conventional fish production with in vitro production. In vitro technology can be used to produce fish tissue that is functionally almost identical to today's products in terms of nutritional value, taste, appearance and odour. Using in-vitro technology, fish can be produced using a fraction of the amount of animals currently produced or caught, thus reducing fishing and counteracting current overfishing. Because in-vitro production takes place under controlled conditions, the products are free of environmental toxins and do not require the use of drugs. Finally, in vitro production makes a significant contribution to animal welfare by eliminating the need for intensive fish farming and the mass killing of fish.
Contact: Petra Kluger
Successfully completed projects - Biomedicine
Lungen-Fettmodell-Medien- Entwicklung eines neuartigen in vitro Lungen-Fettgewebemodells und definierter Zellkulturmedien zur Untersuchung des Einflusses von Adipositas auf Lungenerkrankungen
Adipositas ist eine komplexe Erkrankung mit einer Vielzahl von assoziierten Komorbiditäten, die den Stoffwechsel, zahlreiche Organe und Organsysteme betreffen. In den letzten Jahren, weisen zahlreiche Studien zunehmend auf eine wichtige Bedeutung bei Lungenkrankheiten hin. Der chronisch entzündliche Zustand im adipösen Fettgewebe wird dabei häufig als wichtiger Faktor für eine Wechselwirkung zwischen Fettgewebe und Lunge diskutiert. Es ist aber immer noch unklar, wie das entzündliche adipöse Fettgewebe und pathologische Zustände der Lunge in Verbindung stehen. Aufgrund der erwähnten komplexen Pathologie von Adipositas, ist die Aufklärung über ursächliche Zusammenhänge mit Tierversuchen und Patientendaten häufig kaum möglich. Zudem existiert im Moment kein humanes in vitro Gewebemodell aus Lungen und Fettgewebe, das als Testsystem verwendet werden kann. Hinzu kommt, dass humane in vitro Testsysteme aufgrund der Verwendung von FCS im Medium in ihrer Aussagekraft eingeschränkt sind. Es besteht daher ein zunächst einmal ein dringender Bedarf an definierten Zellkulturmedien um in vitro Testsysteme etablieren zu können, die aussagekräftige und reproduzierbare Ergebnisse liefern.
The skin is the largest organ of the human body and consists of three different layers - epidermis, dermis and subcutis. It represents the first contact area to the environment and forms a barrier against harmful influences. Therefore, the skin is of tremendous interest for various industries like chemistry, pharma or cosmetics and animal experiments are still common in these fields to some extent. Since the ban of animal testing for cosmetics and more strict legal regulations, the need for valid alternatives to animal experiments like in vitro test systems is increasing.
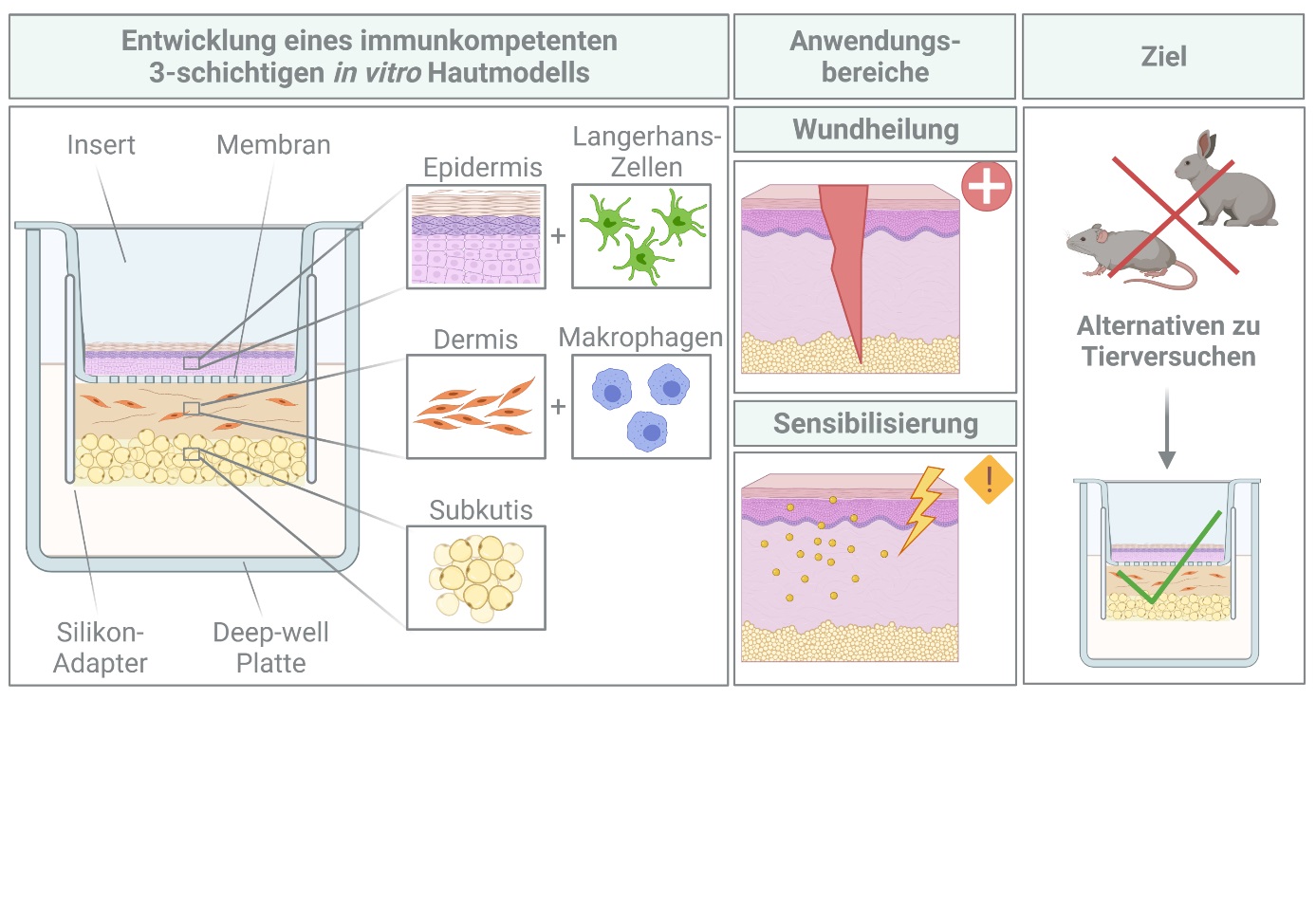
In the already completed previous project “Fat2Skin”, a three-layered human in vitro skin model was successfully established and improved for topical application of irritating substances. The structure of the skin model is comparable to the in vivo situation. In addition, an artificial basal lamina is located between epidermis and dermis, which allows for a tightly sealed epidermal layer optimized for substance testing.
The three-layered in vitro skin model is a versatile tool and can be adapted further for a broad spectrum of applications. In wound healing and sensitization, the immune system of the skin plays a pivotal role in mediating healing and sensitization processes and therefore the addition of immune cells to the in vitro skin model is a prerequisite for simulating a more physiological environment. In the project “Immune-Fat2Skin” an immunocompetent three-layered in vitro skin model is aimed to be developed by incorporating immune cells like Langerhans cells into the epidermal and macrophages into the dermal part of the model. This immunocompetent skin model should then be characterized by histological and immunological techniques and finally validated for wound healing and sensitization studies.
Project duration: 05/2020 until 04/2022
Funding: Ministerium für Ernährung, Ländlichen Raum und Verbraucherschutz Baden-Württemberg
Contact: Petra Kluger, Sophia Nowakowski
This cooperation project between the universities of Aalen and Reutlingen is the research and development of a 6D printing system for biological systems to further develop existing printing and evaluation techniques in tissue engineering. Based on a 6D FDM printer, a suitable printing and measuring unit for biological systems is developed to realize new biomedical products and quality requirements.
For this, free-form web guidance and suitable quality assurance with non-invasive online measurement methods are necessary. The ideal is a process optimization by feedback and the post-control during the process by parallel analysis. The achieved results after two years are the design, development, and testing of a 6D printing demonstrator for biological materials and a prototype of an OCT module for quality assurance.
Overriding goals are:
- flexible automation to achieve increased reproducibility and upscaling for larger biological material assemblies.
- in the future complex biomedical products such as 3D printing of biological material in the operating room directly into the patient, e.g., on a burn wound on the arm.
Project duration: 05/2020 to 05/2022
Funding: Ministry of Science, Research and Arts Baden-Württemberg, Paul Hartmann AG
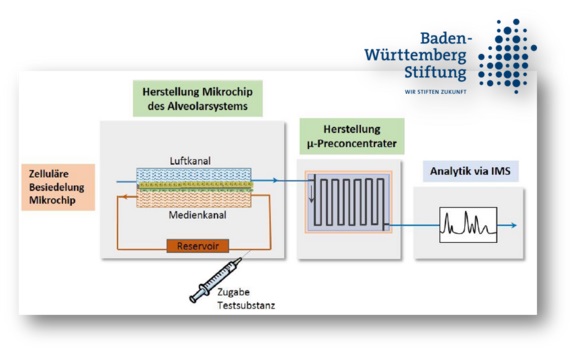
The aim of the interdisciplinary project is to establish a lung microchip for its use in preclinical respiratory gas analysis. The work is a cooperation of the Kluger-Lab and Kemkemer-Lab.
Usually, the dosage of medication is based on body weight or effect. The accompanying measurement of blood concentrations is time-consuming, invasive and expensive. Moreover, the results are only available with after a considerable time delay. The measurement of drug concentrations in the exhaled air represents a promising approach in the individualized medicine and allows to eliminate the previously mentioned disadvantages.
Therefore, an in vitro lung model is urgently needed to investigate the systematic drug transition from "blood" through the tissue into "exhaled air". To build up a physiological and in vivo near alveolar interface, this needs to be simulated using a chip with a membrane and an endothelial-epithelial-co-culture. The focus will be on the formation of a dense and confluent monolayer, as this is a basic prerequisite for the future predictive power of the model. In addition, the model should be long-term stable and persist for at least two weeks in vitro.
Project duration: 03/2018 until 02/2021
Funding: Baden-Württemberg Stiftung
Contact: Petra Kluger, Simon Heine
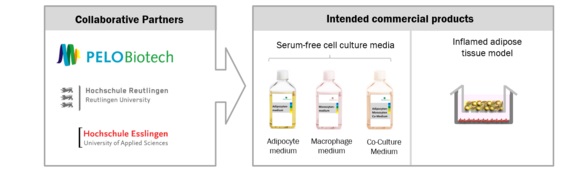
EntdefFett – Setup of an inflamed adipose tissue model with serum-free cell culture media
This project is funded by the Ministry of Research, Science and Art Baden Württemberg and by the European Regional Development Fund.
The main goals within the project are
- The development of serumfree media for the mono- and co-culture of mature adipocytes and a monocytic cell line
- The setup of an 3D adipose tissue model based on mature adipocytes via additive manufacturing
- The setup of an inflamed coculture model in serum-free conditions
Inflamed adipose tissue is associated to many diseases like diabetes or pancreatitis. In vitro models of inflamed adipose tissue can help to reveal the metabolic processes behind the development and manifestation of inflammations in adipose tissue. Furthermore, such models could be used to screen for new drugs addressing this pathological changes.
The aim of this project is the setup of a novel human test system of inflamed adipose tissue. Therefore, the development of defined cell culture media (free of animal-derived serum) is addressed through a process-accompanying analysis.
For the achievement of this ambitious goal, Reutlingen and Esslingen University and PELOBiotech will intensify their application-orientated collaboration. Defined media will developed and tested in adipocyte and monocyte cell culture. Accompanying to this, the process will be analyzed by various parameters at Esslingen University. The possibility to upscale such models with high reproducibility, would substantially simplify their entry to the market (e.g. in pharmaceutical industry). To facilitate the standardized production of inflamed adipose tissue models, the setup of 3D hydrogels including mature adipocytes will be promoted in parallel.
Project duration: 05/2018 until 12/2020
Funding: Ministry of Research, Science and Art Baden Württemberg and by the European Regional Development Fund
Contact: Svenja Nellinger
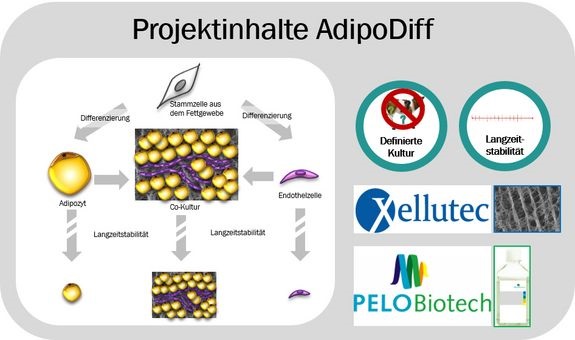
Within this project we wanted to develop and evaluate new cell culture media and matrices for their suitability in differentiation, long term and co culture of adipose stem cells. Thereby, we focused on the differentiation to endothelial cells and mature adipocytes and their coculture, what is of major interest for clinical applications in regenerative medicine or for new in vitro test systems. The project was a cooperation with the biotech companies Bioregeneration and PELOBiotech.
Project duration: 11/2014 until 10/2017
Funding: Bundesministerium für Bildung und Forschung
Contact: Petra Kluger
Clinical treatment of deep and large-scale wounds, e.g. after burning, still remains a challenge. A highly compatible biological material which enhances the cure of the wound with endogenous cells would be preferably.
Aim of the project is the development of such a new material based on tissue specific extracellular matrix (ECM) of patient’s own cells. This native ECM should be modified and optimized using glycobiology. First functional groups are incorporated in the ECM by metabolic glycoengineering. These functional groups can be addressed by biorthogonal ligation reaction and therefore allow the coupling of adhesion supporting oligosaccharides. Further the functional groups can be used for immobilization of the ECM on different materials, e. g. artificial skin. An additional aim is the modification of the ECM with photo-crosslinking polymers to create a material which gel after liquid application and reaches an optimal fill in of the wound.
Project duration: 06/2017 until 06/2020
Funding: Ministerium für Wissenschaft, Forschung und Kunst
Contact: Petra Kluger, Svenja Nellinger
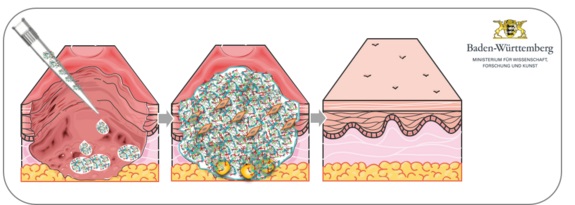
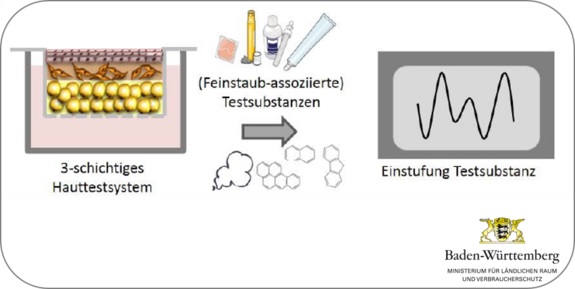
The human skin represents the major contact area to the environment and a barrier towards harmful external influences. In the chemical, cosmetic and pharmaceutic industry the classification of the toxicity of a substance is mostly made by skin reactions. For lack of adequate alternatives still a host of animal experiments are performed. Due to interspecies variances the transferability of the results to the human system is only possible to a limited extend.
Existing cell-based 2-layered (epidermis and dermis) human in vitro skin models represent a good alternative. However also the subcutaneous adipose tissue (subcutis) plays an essential role in storage and transformation of different substances and influences as an endocrine organ the skin reaction. To enhance the reliability and comparability of such tests with human skin a 3-layered human skin model including the subcutis, is urgently needed. The aim to the project is to establish a 3-layered in vitro human skin model and validate it as testing system. Further the established testing system should be used to investigate the influence of fine dust associated substances on the human skin.
Project duration: 11/2017 until 10/2019
Funding: Ministerium für Ländlichen Raum und Verbraucherschutz
Contact: Petra Kluger
Successfully completed projects - cultured meat
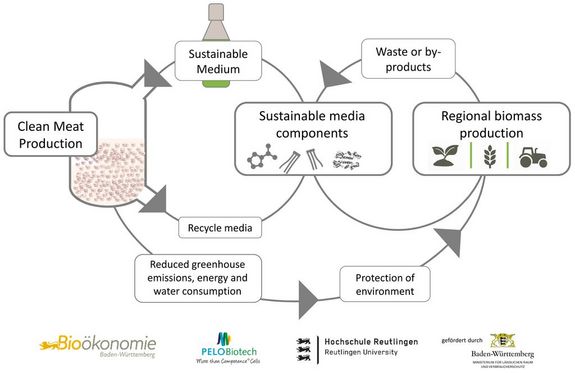
Sustainable nutrient media for the industrial production of clean meat
Laboratory-grown clean meat can meet future nutritional needs while overcoming the environmental problems of conventional meat production and meeting consumer demands through sustainable biotechnological product and process innovations. However, current cultured meat production requires great amounts of animal serum and or cost-intensive supplements. For the sustainable industrial production of fat and muscle cells as a basis for the production of meat from the laboratory, large quantities of resource-saving and at the same time cost-efficient culture media will be required in the near future. For this purpose, regional, already existing biological resources are to be used efficiently. These can be, for example, a wide variety of plant extracts or by-products or waste products from rural biomass production or food industry in Baden-Württemberg.
The objectives of the feasibility study Sustainable Meat are:
1. the identification of cost- or energy-intensive components in the established culture media for the cultivation of animal fat or muscle cells.
2. to identify biomass from rural areas, which is available as a by-product or waste product, for the development of sustainable culture media.
3. the reduction of cost- and energy-intensive components in the established culture media of fat and muscle cells while maintaining cell-typical parameters.
4. preparation of a detailed evaluation catalog to assess the market potentials of different solutions for the production of a sustainable culture medium based on regional biomass.
In order to achieve the intended goals, the Reutlingen University (HSRT) plans a close cooperation of with the company PELOBiotech GmbH (PELO), which has many years of well-founded experience in the field of media development, especially with serum-free media formulations. These culture media should allow the growth of animal fat and muscle cells as well as be sustainable, animal-free and cost-effective to produce.
Project duration: 12/2020 until 08/2021
Contact: Simon Heine, Petra Kluger
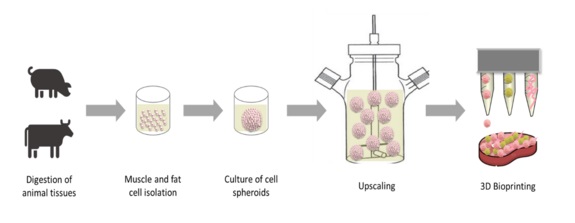
The production of conventional meat in livestock has a great impact on the environment, e.g. on greenhouse gas emissions, water consumption and erosion of the global land area. As the world population is growing, the increasing demand of meat products fuels the further exploitation of resources and further reduces the possibility of welfare-oriented animal husbandry. For these reasons, alternative meat products have to be established. One concept is to develop meat products that are fabricated in the laboratory, in vitro. These products are referred to as Cultured Meat. The strategy hereby is to isolate a small amount of fat and muscle cells from animal tissues and to culture them in vitro to reach an appropriate cell mass. Afterwards, these cells can be processed, for example by bioprinting, to create a Cultured Meat product for consumption.
The aim of this project is to fabricate a product like this by culturing the previously isolated fat and muscle cells as small, round cell aggregates, spheroids. For upscaling, these spheroids will be transferred into a bioreactor system. For further processing, the spheroids can be included in a bioprinting process to produce 3D meat constructs. Therefore, the spheroids should be combined with biomaterials to be printable. The main requirements for suitable biomaterials are: edibility, biocompatibility, printability and they should not contain animal-components. In general, the used components and the whole fabrication process should be environmentally sustainable and inexpensive to obtain an affordable in vitro meat product.
This project is in cooperation with Prof. Dr. Jan Frank and Dr. Monika Bach from the University of Hohenheim.
Project duration: 04/2020 to 04/2023
Funding: Avina Stiftung
Contact: Petra Kluger, Simon Heine
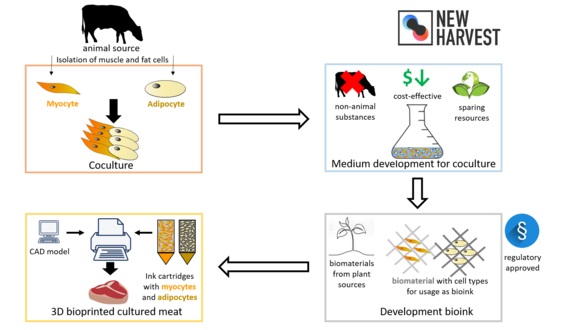
Development of a serum-free co-culture medium, bioinks and a simplified CAD-model for meat printing
This project is funded by the Non-Profit Organisation New Harvest and contains four main work packages. Our aims in this project are:
- Establishment of the isolation and culture of animal derived progenitors of myocytes and adipocytes as well as their coculture
- Development of serum-free medium for muscle and adipose cells
- Development of muscle and adipose cell containing bioinks
- CAD model and printing of simplified 3D units of natural meat structure
Here the approach is pursued to 3D print a meat similar model to imitate the native structure and texture of meat. For the construction of the meat model fat (adipocytes) and muscle cells (myocytes) should be used as basic unit. Therefore, the required and divisible progenitor cells (Adipose derived stem cells (ASCs) and satellite cells) will be isolated from porcine or bovine tissue. Afterwards these both cell types will be stimulated to differentiate into mature adipocytes (from ASCs) and muscle cells (from satellite cells). In the next steps a co-culture medium for both cell types should be developed. For the whole project it is important that all involved materials besides the used cells should be low-cost and from non-animal sources. So, new meat production approaches can be as independent as possible from livestock farming. In detail, it is necessary to exchange commonly applied animal-derived products (like fetal bovine serum, FBS) from culture media with non-animal alternatives. This media adaption helps to further standardize and raise the quality of the product. Beyond that, feasible bioinks based on non-animal materials has to be identified and adapted. These developed “muscle” and “fat” bioinks will be proceeded inside a 3D-bioprinter to fabricate simplified functional 3D-units of meat mimicking the form and texture of native meat tissue. Considering that, computer-aided design (CAD) models, which provide a simplified geometric information regarding shape and composition of muscle and adipose compartment in meat, have to be designed and tested. The claimed results of this project (low-cost defined media, animal-free bioinks, CAD models and printed functional units of meat) will be the basis for bioprinting of meat.
Project duration: 09/2019 until 12/2022
Funding: New Harvest
Contact: Petra Kluger, Jannis Wollschlaeger




![[Translate to English:] [Translate to English:]](/fileadmin/_processed_/5/0/csm_Leikeim_674947ecb5.jpg)


![[Translate to English:] [Translate to English:] Lisa Penberthy](/fileadmin/_processed_/6/2/csm_Penberthy_7872bb5404.jpg)






